Innate Immunogenetics
Despite continuous improvement in the prevention and treatment of human diseases, infection is still one of the leading causes of mortality and morbidity worldwide. Increasing evidence has highlighted the importance of host genetic polymorphism in susceptibility to infectious diseases. The discovery of such polymorphisms may lead to the development of new preventive and predictive strategies for the management of patients at risk to develop severe infection. The aim of our main research is to identify genetic factors associated with the clinical presentation of infectious diseases, and to understand the mechanisms by which these factors influence gene expression and function (precision medicine).
Main Research Topics
Viral Infections
Hepatitis C Virus. The hepatitis C virus (HCV) infection has an estimated worldwide prevalence of 3% and is associated with significant morbidity and mortality. About 30% of infected patients spontaneously clear the virus while othesr evolve towards chronic infection, which is associated with an increased risk of developing fibrosis, cirrhosis and subsequent hepatocellular carcinoma. With the advent of directly acting antiviral agents, a cure rate of >90% can now be achieved. However, due to their important costs, such treatments are restricted to certain areas and patients. Genetic investigations may help development of personalized treatment strategies. Our research in this field is performed in collaboration with the Swiss Hepatitis C Cohort Study (SCCS).
Herpesviruses (HHVs) comprise different DNA viruses that usually cause non-life threatening infections in immunocompetent patients, such as herpes (herpes simplex virus [HSV] 1 and 2), varicella (varicella-zona virus [VZV]) or mononucleosis (Epstein-Barr [EBV and cytomegalovirus [CMV]). In immunocompromised patients, HHVs can be associated with severe and/or life-threatening infections such as CMV disease (that can lead to rejection in solid organ transplant recipients) or disseminated zona. Our research aim at determining host genetic factors associated with susceptibility to these infections. Our research is performed in collaboration with the Swiss Transplant Cohort Study and the HIV Cohort Study.
![Fig 1 immunogenetics polymorphismes [Translate to English:] Fig 1 immunogenetics polymorphismes](/fileadmin/sites/min/images/min-recherche-immunogenetics-polymorphismes-fig1_mod.jpg)
Interferon-λ polymorphisms influence the course of hepatitis C and other viral infections. While numerous studies have shown that polymorphisms within the interferon λ 3/4 (IFNL3/4) locus strongly influence HCV spontaneous or treatment-induced clearance, the mechanisms underlying these associations have remained poorly understood for many years. Recent studies are starting to uncover their role in the differential expression and biology of IFNL3 and 4. Individual ability to clear HCV results from the combination of 2 main polymorphisms (haplotypes) that determine IFNL3 and IFNL4 expression. The active form of IFNL4 does not contribute to clearance, but to a paradoxical response-refractory state in hepatocytes. Recent studies indicate that IFNL3/4 polymorphisms also influence the outcome of infections due to viruses other than HCV, such as CMV, which can cause important complications after organ transplantation (rejection) and in patients with AIDS (blindness).
- Manuel O*, Wojtowicz A*, et al. J Infect Dis. 2015, 211(6):906-14.
- Terczynska-Dyla E*, Bibert S*, Duong F. H*, et al. Nat Comm. 2014; 5:5699.
- Bibert S et al. AIDS 2014; 28(13): 1885-9.
- Bibert S, et al. J Exp Med 2013; 210(6): 1109-16.
- Rauch A et al. Gastroenterology 2010; 138(4): 1338-45.
Invasive Fungal Infections
Aspergillus species can cause invasive pulmonary infections among patients undergoing intensive chemotherapy for the treatment of onco-hematological diseases and/or hematopoietic stem cells or solid organ transplantation. It is still unknown why some patients rapidly develop such infections, while others, under similar immunosuppressive conditions, do not. Candida species can also cause severe infections among hospitalized patients, for instance after long courses of antibiotic treatments or abdominal surgery.
We observed that polymorphisms in different immune genes are associated with increased susceptibility to invasive aspergillosis in transplant recipients (TLR4, IL1-β and PTX3) or invasive candidiasis after complicated abdominal surgery (TNF-α). Our research in this field is performed in collaboration with the Swiss Transplant Cohort Study and the Fungal Infection Network of Switzerland (Funginos).
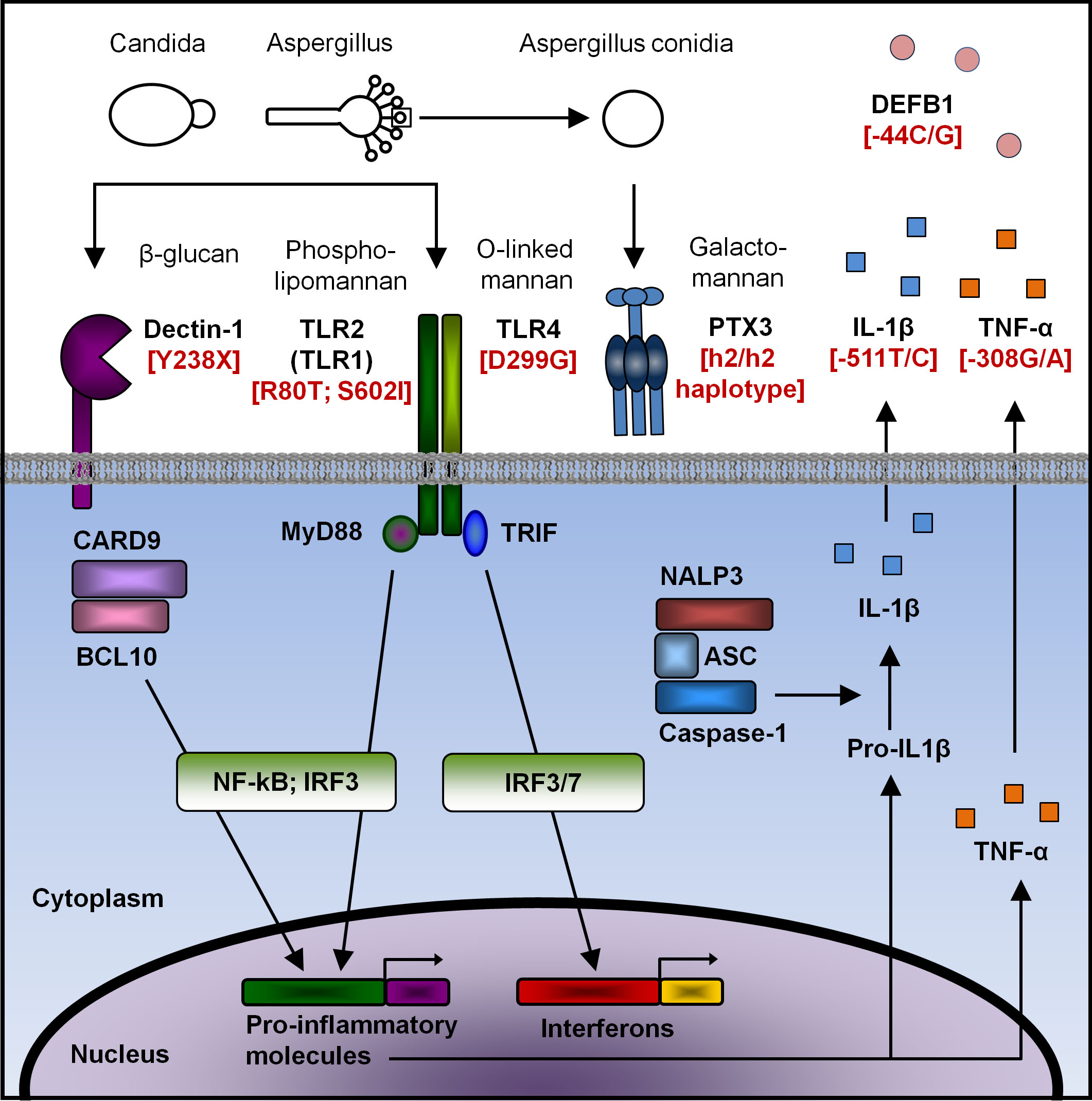
Immunogenetics of invasive fungal infections. Since 1990, several studies have revealed the existence of families of molecules located at the surface or within innate immune cells that can detect specific molecular patterns from microbes. These “pattern recognition receptors”, such as toll-like receptors (TLR), Dectin-1 and pentraxin-3 (PTX3) elicit several signaling pathways, leading to the production of inflammatory molecules, such as cytokines (e.g. IL-1β and TNF-α) and antimicrobial peptides such as defensins (e.g. β-defensin 1 [DEFB1]). These molecules activate other immune cells. This inflammatory cascade leads to the elimination of the offending pathogen. Our study group as well as other investigators showed that variations in immune genes (called polymorphism) can diminish the ability of certain individuals to fight against fungal pathogens, such as Candida or Aspergillus species.
- Wójtowicz A, et al. Clin Infect Dis. 2015; 61(4):619-22.
- Wójtowicz A et al. J Infect Dis. 2015; 211(10):1646-57.
- Wójtowicz A et al. Crit Care Med; 2014; 42(4):e304-8.
- Bochud P.-Y. et al. N Engl J Med 2008;359:1766-77.
Contacts
CHUV
BH 10-537
Rue du Bugnon 46
1011 Lausanne
Switzerland



