Materno-fetal and Obstetrics Research Unit

Since its creation in 2013, Professor David Baud and Dr Milos Stojanov's research team has been active in multiple fundamental and clinical projects in the gynecology, obstetrics and human reproduction fields, thanks to the expertise in maternal-fetal medicine, medical microbiology, bioinformatics, cellular and molecular biology.
Research Topics
Impact of genital bacteria on human reproduction
Bacteria and other microorganisms colonize most of the human body. They are not passive commensals but have a profound influence on the host homeostasis and play a significant role at multiple levels, including protection against pathogens, maturation of the immune system, metabolic pathways, vitamin synthesis and others. It is thus not surprising that a disbalance or dysbiosis of the microbiota has been associated with several adverse outcomes.
The human genital tract is not an exception to bacterial colonization. While the existence of a vaginal microbiota has been well established, other genital sites have been considered sterile environments with a general assumption that bacterial presence is a pathological situation. However, recent studies identified specific patterns of microbiota colonizing these sites, but their role remains poorly explored.
Dysbiosis of the genital microbiota has been linked to the impairment of reproduction and adverse pregnancy outcomes such as preterm birth. Despite this, studies on the impact of bacteria remain scarce and, when available, are descriptive analyses. In particular, more effort is needed towards understanding host-microbial interactions at the molecular level and how microbiota could be shaped to restore homeostasis.
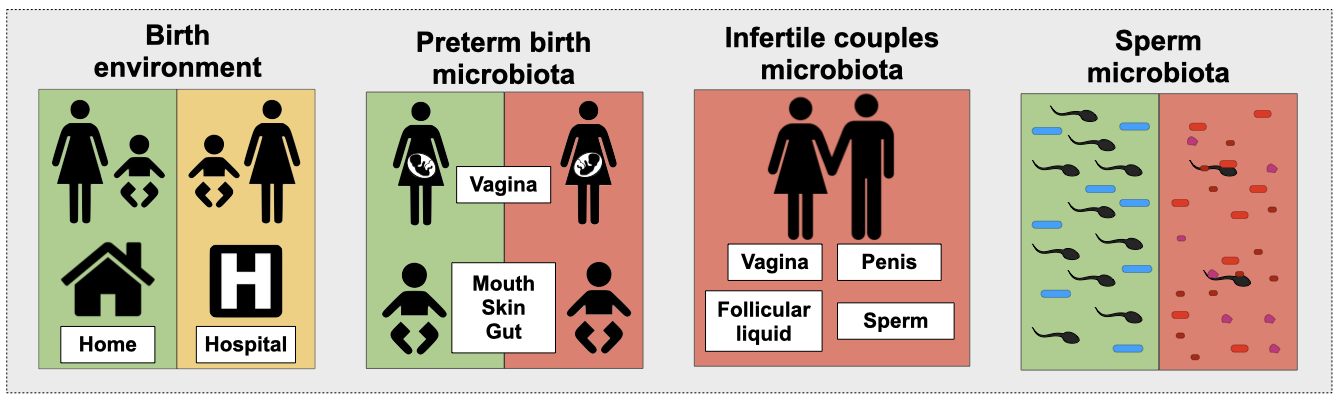
Our research interests span between the early stages of microbial colonisation of the newborns, the impact of the microbiota on preterm births, the interplay between partners’ microbiota and its possible role in infertility.
Related publications:
- Home or hospital birth: the neonatal microbiota perspective. Stojanov M., Das S., Odent M., Engel P., Baud D., 2022/04. The Lancet. Microbe, 3 (4) pp. e247.
- Bacterial Colonization of the Female Upper Genital Tract. Peric A., Weiss J., Vulliemoz N., Baud D., Stojanov M., 2019/07/11. International journal of molecular sciences, 20 (14).
- Sperm Microbiota and Its Impact on Semen Parameters. Baud D., Pattaroni C., Vulliemoz N., Castella V., Marsland BJ, Stojanov M., 2019/02/12. Frontiers in Microbiology, 10 pp. 1-9.
Development of experimental models to evaluate of the impact of genital microbiota on host’s physiology
Once specific bacteria are identified in observational studies (such as 16S rRNA-based community analyses), their positive or negative impact should be further explored and confirmed using animal models or in vitro models of infection/colonization.
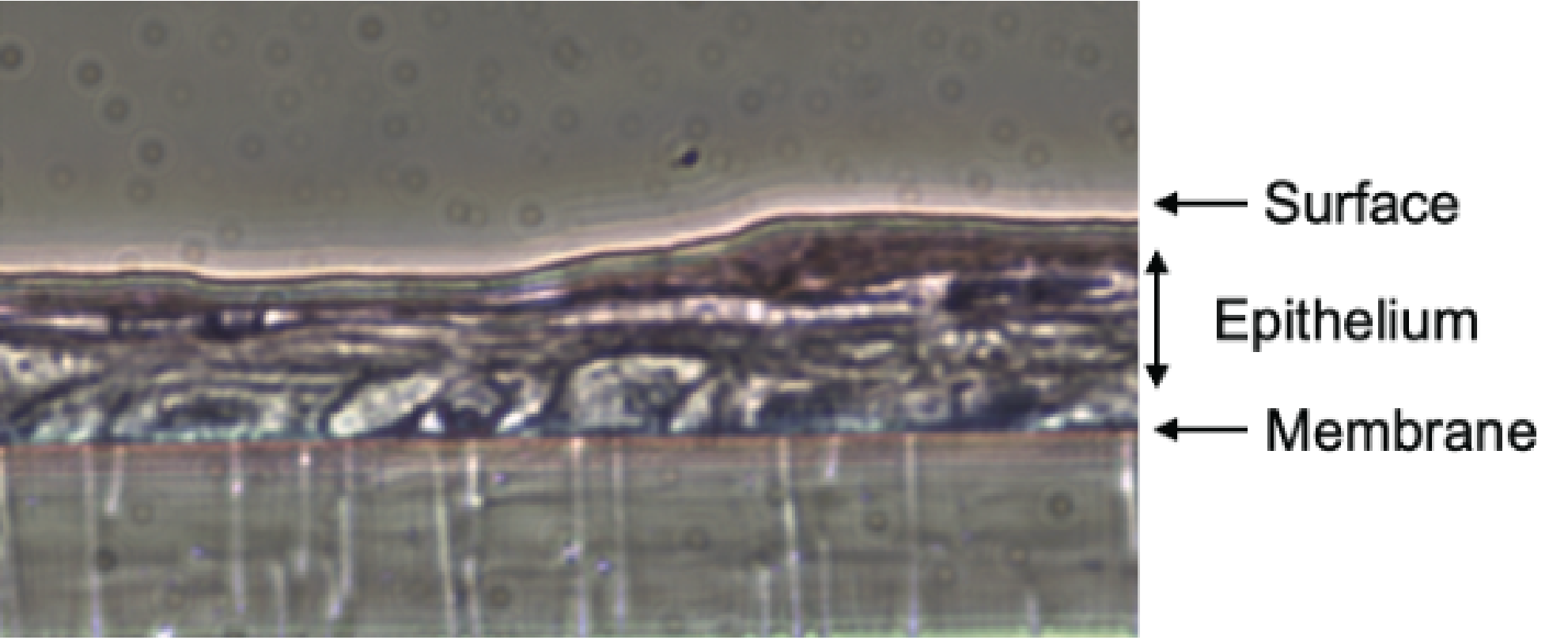
Vaginal microbiota
In the specific case of the vagina, this is not a trivial task as the human female lower genital tract has unique features compared to other mammals. The healthy human vagina is characterized by a low pH (median pH of 4.5 compared to 5.4–7.8 among other mammals) and is colonized mainly by bacteria of the genus Lactobacillus. This is not the case for other animals, including our closest relatives (chimpanzee and gorilla). Consequently, animal models (mainly mouse) used in the past to study bacterial colonization and pathogenesis of the genital tract are not optimal due to these important physiological differences.
Our goal is to develop new in vitro experimental system to study genital infections, dysbiosis and bacterial competition without using animal models. We are currently developing an in vitro vaginal epithelium.
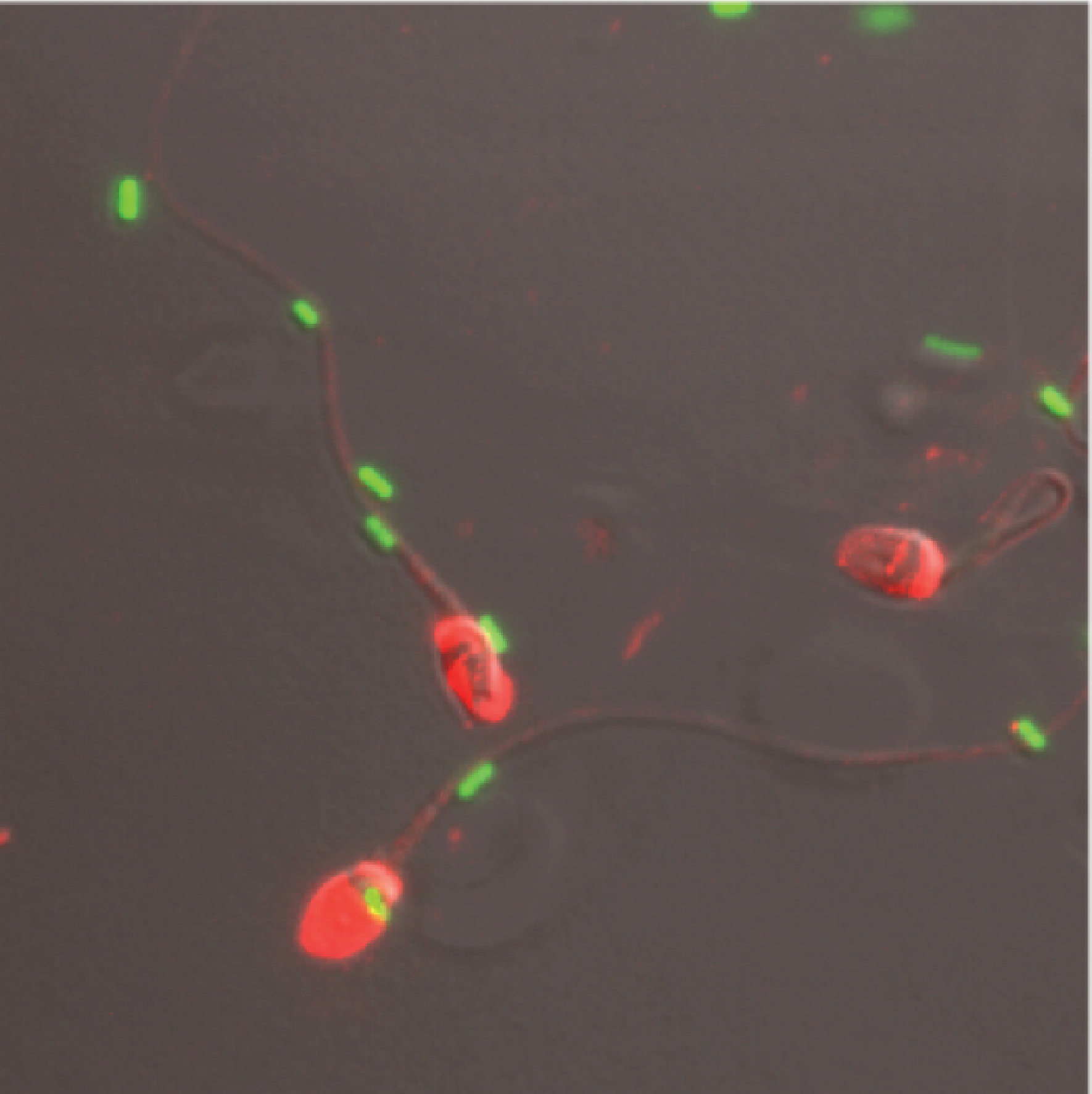
Seminal microbiota
As we have previously reported, seminal fluid may be colonised by a complex community of bacteria. We aim at characterising the effect of the major genital isolates on sperm physiology. We monitor bacteria-spermatozoa interaction using confocal microscopy and transmission electron microscopy. Furthermore, we monitor the impact of bacteria strains on viability, mitochondrial membrane potential (directly linked with sperm motility) and DNA fragmentation (TUNEL assay).
Related publications:
- Waddlia chondrophila, a Chlamydia-related bacterium, has a negative impact on human spermatozoa. Baud D, Vulliemoz N, Ammerdorffer A, Gyger J, Greub G, Castella V, Stojanov M. Human Reproduction. 2018 Jan 1;33(1):3-10.
Pathogenesis of Zika virus and other teratogenic viruses
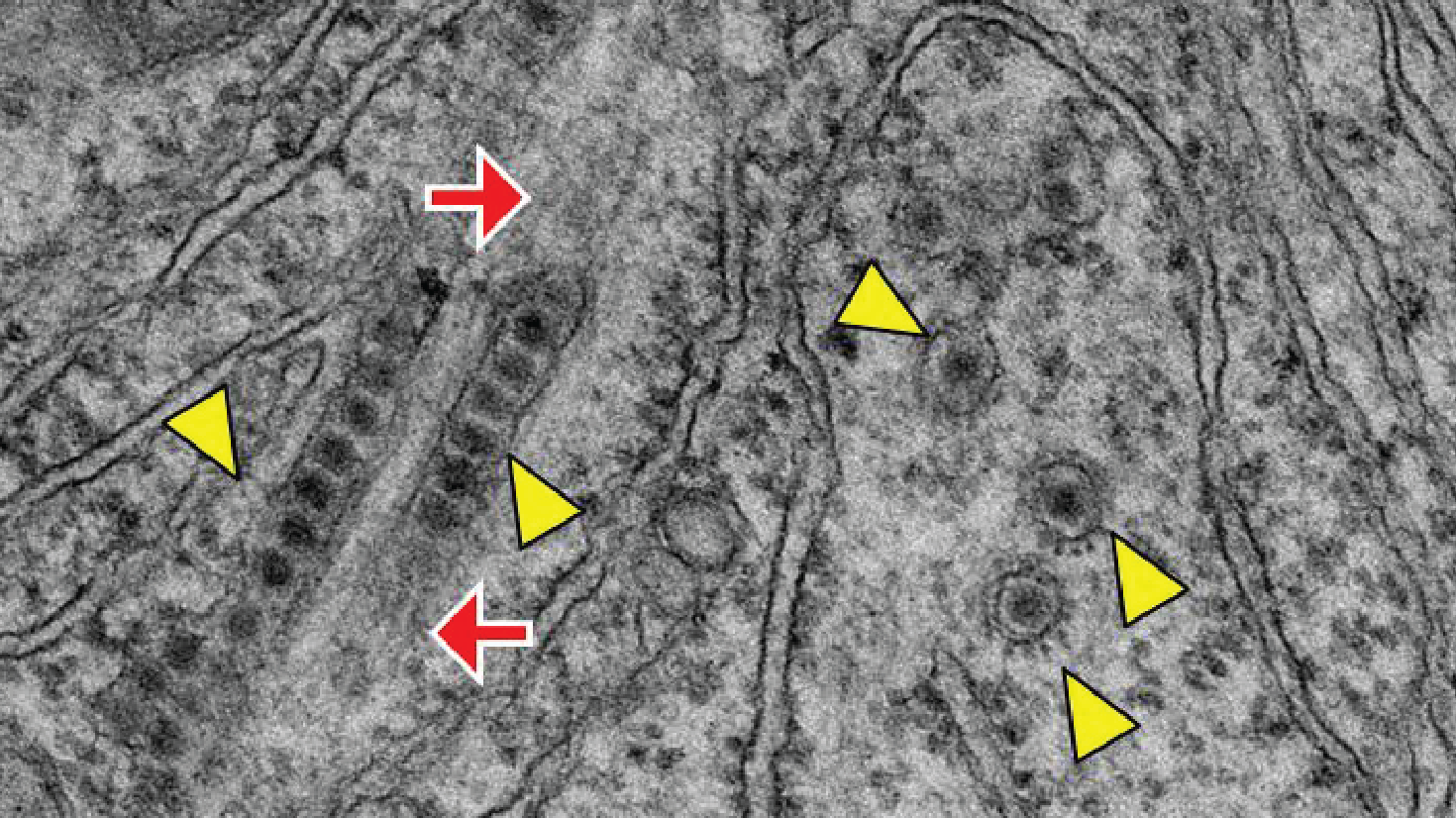
Perinatal viral infections might impact the correct fetal development, leaving important consequences after birth. This is the case for well-known pathogens, like in the case of cytomegalovirus, and emergent pathogens like Zika virus. It is of crucial importance to study the pathogenesis and epidemiology of such viruses in order to envision effective measures to protect both the mothers and their children.
Coupled to clinical studies, our group is investigating the fundamental processes, including the entry of the viruses in host cells and the replication steps of the viruses. A particular importance is given to the ultrastructural analyses of infected cells using electron microscopy.
Related publications:
- Prolonged Maternal Zika Viremia as a Marker of Adverse Perinatal Outcomes. Pomar L., Lambert V., Matheus S., Pomar C., Hcini N., Carles G., Rousset D., Vouga M., Panchaud A., Baud D., 2021/02. Emerging infectious diseases, 27 (2) pp. 490-498.
- Placental infection by Zika virus in French Guiana. Pomar L., Lambert V., Madec Y., Vouga M., Pomar C., Matheus S., Fontanet A., Panchaud A., Carles G., Baud D., 2020/11. Ultrasound in obstetrics & gynecology, 56 (5) pp. 740-748.
- Dengue, Zika and chikungunya during pregnancy: pre- and post-travel advice and clinical management. Vouga M., Chiu Y.C., Pomar L., de Meyer S.V., Masmejan S., Genton B., Musso D., Baud D., Stojanov M., 2019/12/23. Journal of travel medicine, 26 (8) pp. 1-13.
- A critical analysis of the neurodevelopmental and neurosensory outcomes after 2 years for children with in utero Zika virus exposure. Vouga M., Pomar L., Panchaud A., Musso D., Baud D., 2019/11. Nature medicine, 25 (11) pp. 1641-1642.
- Zika Virus Infection - After the Pandemic. Musso D., Ko A.I., Baud D., 2019/10/10. The New England journal of medicine, 381 (15) pp. 1444-1457.
Pathogenesis of Chlamydia trachomatis
C. trachomatis is the most commonly reported sexually transmitted bacterial infection worldwide, with more than 100 million new cases annually, 90% being asymptomatic. However, infection may translate into significant complications such as miscarriage, preterm birth, neonatal pneumonia, tubal infertility, ectopic pregnancy, pelvic inflammatory disease (PID), blindness and urethritis.
Our group is currently focused on the initial steps of chlamydial infection, which, despite the notoriety of this bacterium, remains understudied. Our main goal is to identify all the cellular receptors exploited by this pathogen to penetrate into host cells.
Related publication:
Pathogenesis of Chlamydia-like bacteria
Waddlia chondrophila, an emerging pathogen associated with miscarriages
Approximately 25% of women will experience at least one miscarriage during their life. Despite a large number of possible investigations, a cause of the miscarriage is identified in less than 50% of the cases. Intracellular bacteria, which are not routinely investigated in clinical practice, might represent possible agents of miscarriages of unknown etiology. Listeria monocytogenes, Chlamydia trachomatis and Coxiella burnetii are known intracellular bacteria associated with adverse pregnancy outcome. Recently, several studies identified Waddlia chondrophila, an emerging intracellular bacterium belonging to the order of Chlamydiales, to be strongly associated with adverse pregnancy outcomes both in women and animals. W. chondrophila positive serologies as well as PCR and immunohistochemistry (both in the genital tract and placenta) were more likely to be present in women with miscarriages.
Our group is currently investigating:
- The mode of infection of W. chondrophila infection during pregnancy;
- The immune response against W. chondrophila in women who experienced miscarriage compared to women with uneventful pregnancies;
- The immunological mechanisms related to W. chondrophila induced miscarriage.
Related publications:
- Chlamydia trachomatis and chlamydia-like bacteria: new enemies of human pregnancies. Ammerdorffer A, Stojanov M, Greub G, Baud D. Curr Opin Infect Dis. 2017 Jun;30(3):289-296.
- Waddlia chondrophila and Male Infertility. Baud D., Vulliemoz N., Zapata MVM, Greub G., Vouga M., Stojanov M., 2020/01/17. Microorganisms, 8 (1).
- Impact of Waddlia chondrophila infection on pregnancy in the mouse. Baud D., Ammerdorffer A., Buffe Y., Vouga M., Greub G., Stojanov M., 2020/01. New microbes and new infections, 33 p. 100619.
- Waddlia chondrophila induces systemic infection, organ pathology, and elicits Th1-associated humoral immunity in a murine model of genital infection. Vasilevsky S., Gyger J., Piersigilli A., Pilloux L., Greub G., Stojanov M., Baud D., 2015. Frontiers In Cellular and Infection Microbiology, 5 p. 76.
- Role of Waddlia chondrophila placental infection in miscarriage. Baud D., Goy G., Osterheld M.C., Croxatto A., Borel N., Vial Y., Pospischil A., Greub G., 2014. Emerging Infectious Diseases, 20 (3) pp. 460-464.
Contact
Ch. des Boveresses 155
1066 Epalinges



